Two hours in a hospital chair every three weeks, a sterile bag prepared under specialist conditions, a cannula threaded into a vein. For a decade, this was the routine for thousands of NHS patients receiving pembrolizumab — one of the most successful cancer drugs ever developed.
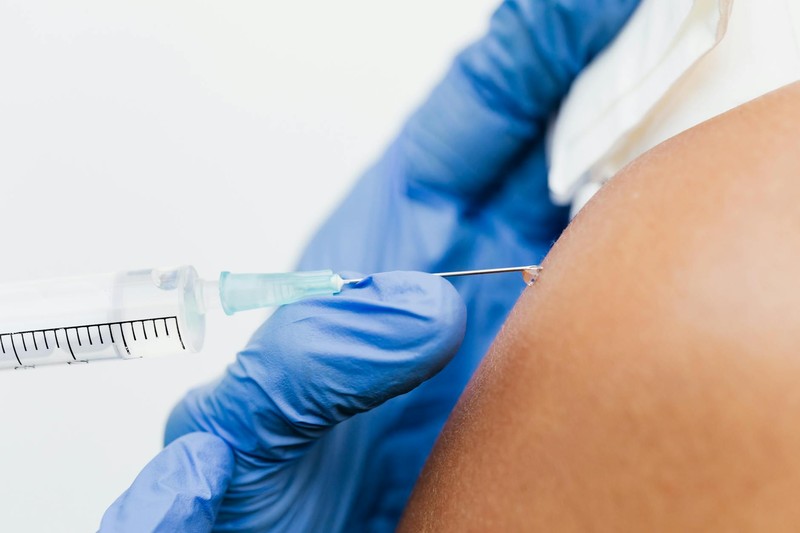
Now the same treatment takes sixty seconds.
The NHS has begun rolling out an injectable form of pembrolizumab, brand name Keytruda, that replaces the intravenous drip with a quick jab under the skin. Around 14,000 patients in England who start pembrolizumab therapy each year are expected to benefit, and the change could save the health service more than 100,000 hours of preparation and treatment time annually, according to NHS England.
How it works
Pembrolizumab is a type of immunotherapy — a treatment that doesn’t attack cancer directly but instead removes the brakes from the patient’s own immune system.
Cancer cells produce proteins that send a “stop signal” to immune cells, effectively hiding behind what some scientists describe as an “invisibility cloak.” Pembrolizumab blocks a protein called PD-1, which acts as one of those brakes. Once PD-1 is neutralised, the immune system can recognise and destroy cancer cells it previously ignored.
That discovery earned scientists James Allison and Tasuku Honjo the Nobel Prize for Medicine in 2018. Since then, pembrolizumab has become the world’s best-selling prescription drug, with global sales of $30 billion in 2025 alone, according to the BBC.
The injectable version covers 14 cancer types in the UK, including lung, breast, head and neck, and cervical cancers. It is administered every three weeks as a one-minute injection or every six weeks as a two-minute injection, depending on the diagnosis.
A different kind of hospital visit
Shirley Xerxes, an 89-year-old from St Albans, was one of the first patients to receive the new jab at Mount Vernon Cancer Centre.
“I can’t believe how little time it took,” she said. “I was only in the chair for a matter of minutes instead of an hour or more. It’s made such a difference and gives me more time to live my life, including spending more time gardening.”
The shift is more than a convenience. The IV version requires hospital pharmacy teams to prepare bags under sterile conditions — a time-consuming process that ties up specialist staff and clinic space. The injectable form comes ready to administer, freeing up both physical capacity and clinical hours.
“If we can do this in a much shorter period of time, it frees up space in our chemotherapy units and means we can start thinking about giving treatment in the community and away from hospitals,” said Professor Peter Johnson, NHS national clinical director for cancer.
The bigger picture
The rollout is the latest sign of a broader shift in cancer care delivery. Last year, up to 15,000 patients became eligible for a similar injectable form of nivolumab, another immunotherapy drug. Together, the two jabs now cover nearly 30 cancer types on the NHS.
In the United States, the FDA has approved the subcutaneous version — branded Keytruda Qlex — for 38 cancers. Manufacturer Merck, known as MSD outside the US, says it will price the injection “at parity” with the IV version.
Not everyone is celebrating unreservedly. In the US, critics including Senator Elizabeth Warren have accused Merck of introducing the injectable form — covered by separate patents — partly to shield the drug from cheaper generic competition. The original drug’s patents expire in 2028 in the US and 2031 in Europe. Merck says the jab offers “meaningful benefits” for hospitals and patients.
James Richardson, national specialty adviser for cancer drugs at NHS England, called the change “a win-win innovation.” Michelle Mitchell, chief executive of Cancer Research UK, said the new form would allow people to spend more time living outside hospital — though she added that at a time of severely limited NHS capacity, the government must ensure proven treatments “reach patients quickly and fairly.”
For patients like Shirley Xerxes, the economics are less pressing than the afternoon she got back. She described the experience as unbelievable. More time for gardening. Less time in a chair.
Sources
- Thousands could benefit from cancer jab that cuts treatment time to minutes — BBC News
- ‘1-minute’ immunotherapy jab rolled out on NHS for tens of thousands with cancer — NHS England
- Thousands of cancer patients in England to benefit from new immunotherapy jab — The Guardian
- As Exclusivity Loss Looms, Merck Wins Subcutaneous Approval for Keytruda — BioSpace

Discussion (9)